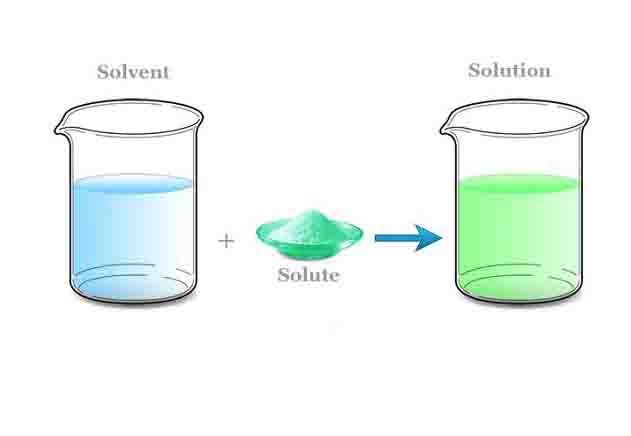
Essentially, a solute is a substance that can be dissolved by a solvent to form a solution. There are many types of solutes. It can be a gas, a liquid, or a solid. Solvents break up solute molecules and distribute them evenly, thereby dissolving them. As a result, a homogeneous mixture, or solution, is created.
Concentrations of solutes in solution are used to measure their concentration. A solute’s concentration is the amount of solute divided by the total volume of the solution. Depending on how strong a solvent is and how easily molecules of a solute separate, a solvent can dilute various amounts of solute. Solubility refers to a solute’s ability to dissolve in a solvent.
Examples of Solute
Salt in Water
When you dump a spoon full of salt into a glass of water, you are creating a solution. The solute is the salt, or NaCl. The solvent is water, or H2O. The water molecules are negatively charged on the oxygen atoms and positively charged on the hydrogen atoms. Salt is an ionic compound, which consists of two ions: Na+ and Cl–.
The negative oxygen atoms attract the positive sodium (Na+), and the positive hydrogen atoms attract the negative chlorine atoms (Cl–). The attraction between the different molecules pulls the solute apart at a molecular level, and suspends it evenly throughout the water.
An important factor in how fast the solute will dissolve is the surface area of solute exposed. If coarse salt is used, less surface area is exposed and it will take longer for the same amount of salt to dissolve.
A finer salt allows many more ions to be exposed to water, and the solute gets diffused through the water faster. Eventually the salt can no longer be seen on the bottom of the glass because it is evenly distributed throughout the glass.
A similar process happens with sugar, but the sugar molecules are not the same as salt molecules. Instead of being an ionic compound, the sugar molecules are slightly polar. The molecule of sugar has many OH groups, which create natural dipoles. These positive and negative areas interact with the positive and negative areas of the water molecules, and the solute molecule are torn apart.
Just as salt is diffused across a solution, sugar can also be evenly distributed in a cell. This is important for many cellular functions, such as producing energy and larger molecules. Other times, cells must actively transport certain molecules out of the cytosol, to avoid upsetting the pH balance.
Oxygen in Seawater
An example of a gaseous solute is oxygen. Every fish in the ocean, from the strange creatures in the deepest parts of the ocean to the common coral-dwelling fish that scuba divers love, rely on oxygen dissolved in the water to live. The oxygen, which exists as O2, is a polar molecule.
As such, the polar water molecules have a natural tendency to attract the oxygen. As the waves mix air into the ocean and the surface of the ocean and atmosphere interact, oxygen is dissolved into the water. The process of diffusion carries the oxygen through the water column, delivering oxygen to organisms throughout the ocean.
In some situations the organisms in the ocean can use the oxygen in the water faster than it can be diffused into the water. This can happen when excess nutrient runoff from humans runs into the ocean. The nutrients, which are another solute in water, allow huge algal blooms to grow. These blooms contain far too many algae.
The algae in the lower layers start to die, and bacteria start to consume them. Between the algae and the bacteria, all of the oxygen gets used up. This creates a dead zone in the water column. If fish start to swim through this column, they could suffocate from lack of oxygen.
Protons in the Cytosol
Organisms of all kinds must regulate the amount of solutes in their cells, to maintain proper cell functions. The acidity of cells is based in part on the number of hydrogen ions (H+), or protons, found in the solution of cytosol. The protons are attracted to the oxygen atoms of water, because they are electronegative.
The protons as a solute serve a very important function in cells. While water is able to diffuse through a cellular membrane via osmosis, hydrogen atoms cannot breach the membrane. The concentration gradient creates a potential force that can be used to move other substances. This is known as proton motive force and is used to move a wide variety of substances through the cellular membrane.
Related Biology Terms
- Solution – A homogenous mixture of solute and solvent.
- Solvent – A substance which can dissolve a solute.
- Concentration Gradient – The difference between the concentrations of solute in two areas of solution.
- Solubility – The ability of a given solute to dissolve in a solvent.
FAQ’s
A solute is a substance that is dissolved in a solvent to form a solution. It can be a solid, liquid, or gas and is typically present in smaller amounts than the solvent.
The interactions between solutes and solvents can be classified into four categories: ion-dipole, dipole-dipole, hydrogen bonding, and London dispersion forces. These interactions determine the solubility of a solute in a given solvent and the properties of the resulting solution.
The concentration of a solute in a solution determines the properties of the solution, such as its density, boiling and freezing points, and osmotic pressure. Increasing the concentration of a solute in a solvent will increase the solution’s properties while decreasing the concentration will have the opposite effect.
Common solutes include salt, sugar, and acids, while common solvents include water, alcohol, and acetone. In biological systems, solutes such as glucose, amino acids, and electrolytes are dissolved in water to form intracellular and extracellular fluids. In industry, solvents such as petroleum-based products and organic solvents are commonly used to dissolve and extract solutes from raw materials.